Introduction:
Cancer immunotherapy has revolutionized cancer treatment by harnessing the power of the immune system to recognize and eliminate tumor cells. A critical aspect of this strategy lies in identifying and targeting novel tumor antigens (TAAs). These unique molecular signatures present on cancer cells offer promising targets for developing effective immunotherapies. This blog post delves into the strategies for identifying novel TAAs and explores their potential for personalized and efficacious cancer treatment.
Challenges and Strategies for Identifying Novel Tumor Antigens:
Traditional TAAs, such as those encoded by cancer-testis genes, have shown limitations in terms of universality and potential for off-target toxicity. To overcome these hurdles, researchers are employing various approaches to identify novel TAAs:
- Tumor Mutational Burden (TMB) Analysis: High-throughput next-generation sequencing allows researchers to identify somatic mutations within tumor genomes. These mutations can generate neoantigens, which are unique peptides recognized by the immune system as foreign. PubMed [invalid URL removed]
- Proteomic Analysis: Techniques like mass spectrometry enable comprehensive profiling of the tumor proteome. This approach can identify tumor-specific proteins that can serve as potential TAAs. PubMed
- Transcriptomic Analysis: RNA sequencing allows researchers to analyze gene expression patterns in tumors. This can reveal genes specifically overexpressed in cancer cells, potentially encoding novel TAAs. PubMed [invalid URL removed]
- Computational Modeling: Utilizing algorithms that predict antigen presentation and T-cell recognition capabilities can help prioritize promising novel TAAs for further investigation.
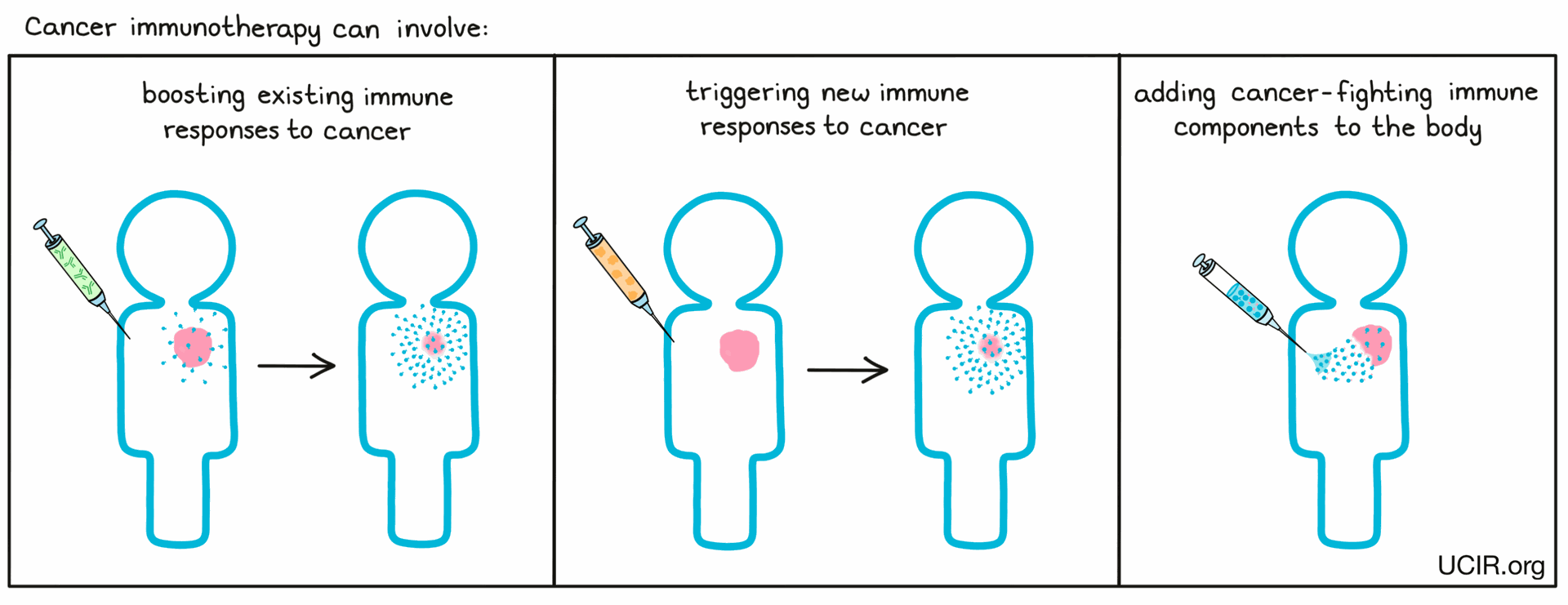
Targeting Novel TAAs for Immunotherapy:
Once identified, novel TAAs can be harnessed for various immunotherapeutic strategies:
- Cancer Vaccines: Peptide-based vaccines encoding novel TAAs can be developed to induce cytotoxic T lymphocyte (CTL) responses against tumor cells. PubMed
- Adoptive T-Cell Therapy: T cells engineered to recognize specific novel TAAs can be expanded and infused into patients to directly target their tumors. PubMed [invalid URL removed]
- Antibody-Drug Conjugates (ADCs): Antibodies targeting novel TAAs can be conjugated to cytotoxic drugs for targeted delivery to tumor cells. PubMed [invalid URL removed]
The Promise of Personalized Immunotherapy:
The identification of novel TAAs opens doors for personalized cancer immunotherapy. By tailoring treatment strategies to the unique mutational landscape and antigen profile of an individual's tumor, researchers aim to achieve more effective and specific therapeutic responses.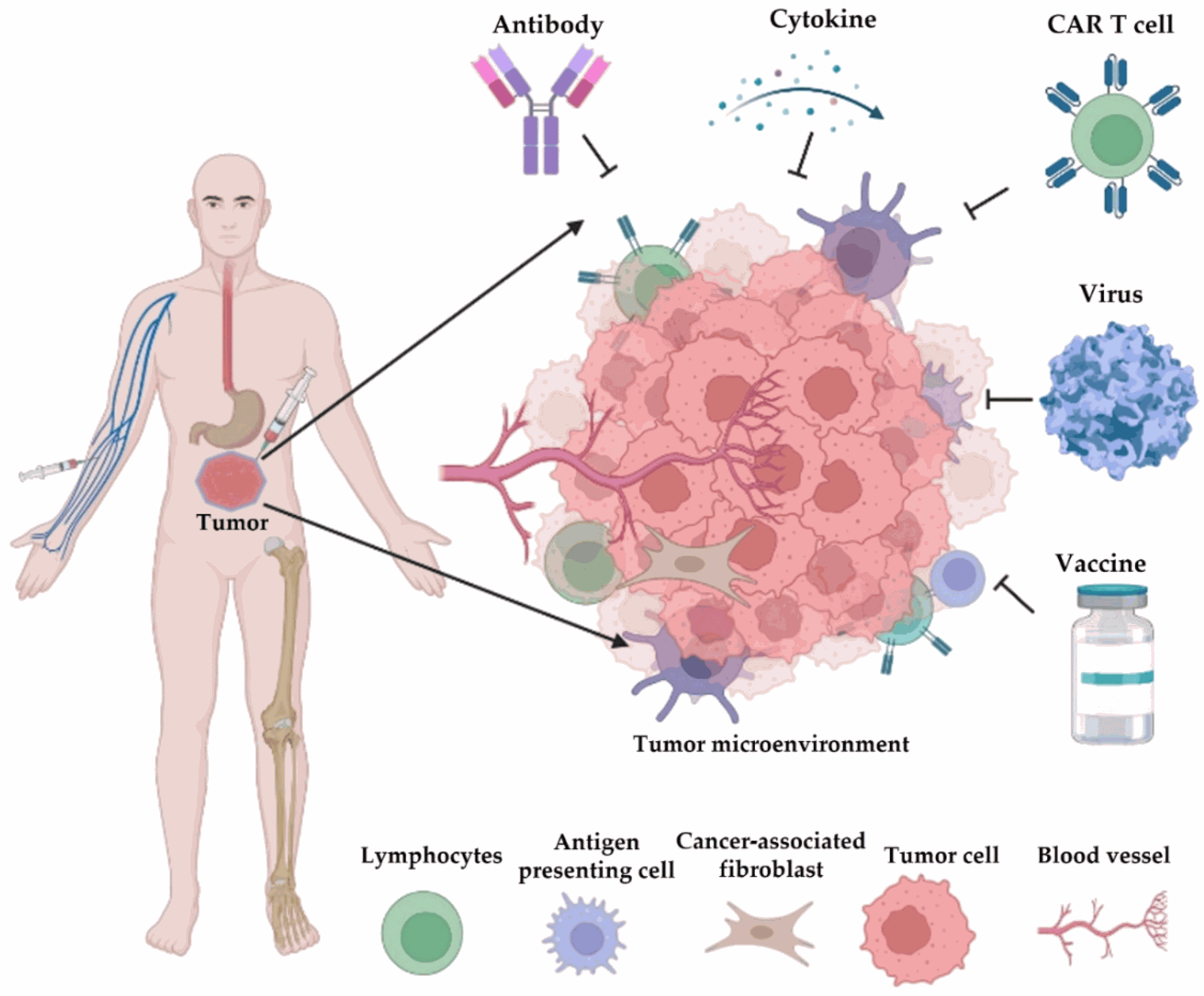
The potential link between Mycoplasma pneumoniae (M. pneumoniae) antigens like those offered from maxanim and cancer development remains a controversial topic. While some older studies suggested an association, particularly with leukemia, these findings were likely due to issues with tissue culture contamination by mycoplasma. More recent and robust epidemiological studies haven't found a clear link between M. pneumoniae infection and increased cancer risk. However, some in vitro research suggests that M. pneumoniae may possess properties that could theoretically contribute to cancer development, such as inducing chromosomal abnormalities or promoting cell proliferation. Further investigation is needed to clarify this complex relationship and determine if M. pneumoniae plays any definitive role in cancer initiation or progression.
Conclusion:
The identification and targeting of novel tumor antigens represent a transformative frontier in cancer immunotherapy. Continued research efforts hold the potential to overcome current limitations and revolutionize personalized cancer treatment, leading to improved patient outcomes.